Chemical Kinetics and Nuclear ChemistryHard
Question
With the help of following reactions, arrange metals A,B,D and E in decreasing order of their reactivity :-
(i) B + ANO3 → BNO3 + A
(ii) A + HCl → ACl +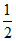 H2
H2
(iii) D + ECl → DCl + E
(iv) D + HNO3 → No reaction
(i) B + ANO3 → BNO3 + A
(ii) A + HCl → ACl +
(iii) D + ECl → DCl + E
(iv) D + HNO3 → No reaction
Options
A.B > D > E > A
B.B > A > D > E
C.E > D > B > A
D.None of these
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate law for a reaction between the substances A and B is given by rate = K[A]n [B]m. On doubling the concentration ...A certain zero order reaction has k = 0.025 M s-1 for the disappearance of A. What will be the concentration of A after ...In a reaction, A + B → Product, rate is doubled when the concentration of B is doubled, and rate increases by a fa...For the consecutive first-order reactions: $A\overset{\quad K_{1}\quad}{\rightarrow}B\overset{\quad K_{2}\quad}{\rightar...An aqueous solution is 1.00 molal in KI. Which change will cause the vapour pressure of the solution to increase ?...