Chemical Kinetics and Nuclear ChemistryHard
Question
Which of the following statements are correct about half-period.
Options
A.It is proportional to initial concentration for zeroth order
B.Average life = 1.44 half-life for first order reaction.
C.Time of 75% completion of reaction is thrice of half-life(initial half life) period in second order reaction
D.99.9% reaction takes place in 100 minutes for the case when rate constant is 0.0693 min-1
Solution
(a) t1/2 ∞ 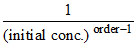
(b) tavg =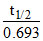
(c) For second order reaction t1/2 =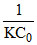
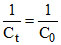 + Kt
+ Kt
At t = t75% Ct =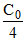
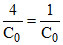 + Kt75%
+ Kt75%
t75% =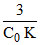
t75% = 3 × t1/2
(b) tavg =
(c) For second order reaction t1/2 =
At t = t75% Ct =
t75% =
t75% = 3 × t1/2
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
A reaction of first-order completed 90% in 90 minutes, hence, it is completed 50% in approximately :...Which one of the following statement is wrong regarding molecularity of a reaction?...If the fermentation of sugar in an enzymatic solution that is 0.12 M, the concentration of the sugar is reduced to 0.06 ...X-Rays are emitted during...For the non-stoichiometre reaction 2A + B → C + D, the following kinetic data were obtained in three separate expe...