Chemical Kinetics and Nuclear ChemistryHard
Question
If the fermentation of sugar in an enzymatic solution that is 0.12 M, the concentration of the sugar is reduced to 0.06 M in 10 h and to 0.03 M in 20 h. What is the order of the reaction ?
Options
A.1
B.2
C.3
D.0
Solution
In 1st order Rxn, decreases in % of concentration same in same interval of Time
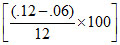 = 50%
= 50%
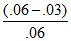 × 100 = 50%
× 100 = 50%
so reaction must be of first order.
so reaction must be of first order.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Identify compound which give fastest dehydration ?...Which of the following is a lyophobiccolloidal solution?...The rate constant of a reaction depends upon...An aqueous solution is 1.00 molal in KI. Which change will cause the vapour pressure of the solution to increase ?...The rate constant for the reaction N2O5 → 4NO2 + O2 is 3.0 × 10−5 s−1. If the rate of reaction is 2.4 × 10−5 mol litre−1...