JEE Main | 2014Chemical Kinetics and Nuclear ChemistryHard
Question
For the non-stoichiometre reaction 2A + B → C + D, the following kinetic data were obtained in three separate experiments, all at 298 K.
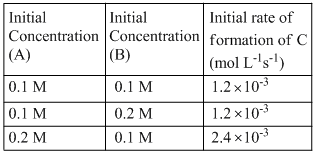
The rate law for the formation of C is

The rate law for the formation of C is
Options
A.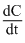 = k[A][B]
= k[A][B]
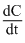 = k[A][B]
= k[A][B]B.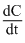 = k[A]2[B]
= k[A]2[B]
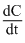 = k[A]2[B]
= k[A]2[B]C.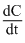 = k[A][B]2
= k[A][B]2
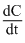 = k[A][B]2
= k[A][B]2D.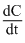 = k[A]
= k[A]
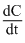 = k[A]
= k[A]Solution
2A + B → C + D
Rate of Reaction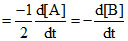
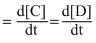
Let rate of Reaction = k[A]x[B]y
Or,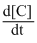 = k[A]x[B]y
= k[A]x[B]y
Now from table,
1.2 × 10-3 = k [0.1]x[0.1]y .......(i)
1.2 × 10-3 = k [0.1]x[0.2]y .......(ii)
2.4 × 10-3 = k [0.2]x[0.1]y .......(iii)
Dividing equation (i) by (ii)
⇒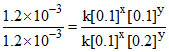
⇒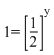
⇒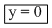
Now Dividing equation (i) by (iii)
⇒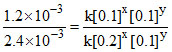
⇒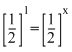
⇒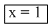
Hence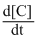 = K[A]1[B]0.
= K[A]1[B]0.
Rate of Reaction
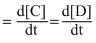
Let rate of Reaction = k[A]x[B]y
Or,
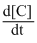 = k[A]x[B]y
= k[A]x[B]yNow from table,
1.2 × 10-3 = k [0.1]x[0.1]y .......(i)
1.2 × 10-3 = k [0.1]x[0.2]y .......(ii)
2.4 × 10-3 = k [0.2]x[0.1]y .......(iii)
Dividing equation (i) by (ii)
⇒
⇒
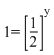
⇒
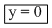
Now Dividing equation (i) by (iii)
⇒
⇒
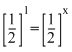
⇒
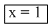
Hence
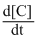 = K[A]1[B]0.
= K[A]1[B]0.Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which of the following reaction(s) is/are of the first order ?...Megasporangium is equivalent to :...Bi210 has a half - life of 5days. The time taken, for of a sample to decay, is :...At 370 K a gaseous reaction X → 2Y + Z is found to be first order. Starting the reaction from pure X the total pre...An α-particle is given out by 92X238, the new species Y would be :...