Chemical Kinetics and Nuclear ChemistryHard
Question
The bromination of acetone that occurs in acid solution is represented by
CH3COCH3 (aq) + Br2 (aq) → CH3COCH2 Br (aq) + HBr (aq)
Given :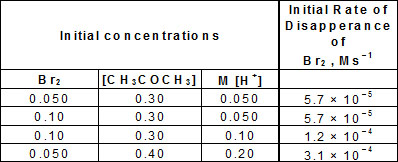
Based on the above data, rate law is :
CH3COCH3 (aq) + Br2 (aq) → CH3COCH2 Br (aq) + HBr (aq)
Given :
Based on the above data, rate law is :
Options
A.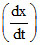 = k [CH3COCH3] [Br2] [H+]
= k [CH3COCH3] [Br2] [H+]
B.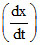 = k [CH3COCH3] [H+]
= k [CH3COCH3] [H+]
C.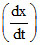 = k [CH3COCH3] [Br2]
= k [CH3COCH3] [Br2]
D.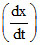 = k [CH3COCH3] [Br2] [H+]2
= k [CH3COCH3] [Br2] [H+]2
Solution
According to above data.
suppose rate law,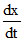 = k [CH3COCH3]a [Br2]b [H+]c
= k [CH3COCH3]a [Br2]b [H+]c
by data (i) and (ii) conc. of [Br2] increased 2 times and conc. of [CH3COCH3] and [H+] constant rate remains same. Then zero order wrt Br2.
Similarly conc. of [H+] increased 2 times rate increased 2 times, so first order wrt H+.
Similarly 1st order wrt CH3COCH3
Then,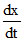 = k [CH3COCH3] [H+]
= k [CH3COCH3] [H+]
suppose rate law,
by data (i) and (ii) conc. of [Br2] increased 2 times and conc. of [CH3COCH3] and [H+] constant rate remains same. Then zero order wrt Br2.
Similarly conc. of [H+] increased 2 times rate increased 2 times, so first order wrt H+.
Similarly 1st order wrt CH3COCH3
Then,
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For irreversible elementary reactions in parallel $A\overset{\quad K_{1}\quad}{\rightarrow}R\text{ and }A\overset{\quad ...A reaction which is of first order w.r.t reactant A, has a rate constant 6 min-1. If we start with [A] = 0.5 mol L-1, wh...For the non-stoichiometre reaction 2A + B → C + D, the following kinetic data were obtained in three separate expe...The correct increasing order of basic strength for the following compounds is :...For the non-equilibrium process, A + B → products, the rate is first-order w.r.t. A and second-order w.r.t. B. If ...