Chemical Kinetics and Nuclear ChemistryHard
Question
A reaction which is of first order w.r.t reactant A, has a rate constant 6 min-1. If we start with [A] = 0.5 mol L-1, when would [A] reach the value of 0.05 mol L-1
Options
A.0.384 min
B.0.15 min
C.3 min
D.3.84 min
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The size of colloidal particle is...The photon of hard gamma radiation knocks a proton out of nucleus to form...For the non-stoichiometre reaction 2A + B → C + D, the following kinetic data were obtained in three separate expe...Which of the following is incorrect about freundlich adsorption isotherm ?...In a certain reaction, 10% of the reactant decomposes in one hour, 20% in two hours, 30% in three hours, and so on. The ...
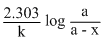
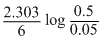 = 0.384 min.
= 0.384 min.