ElectrochemistryHard
Question
Calculate the cell EMF in mV for
Pt | H2 (1atm) | HCl (0.01 M) | AgCl(s) | Ag(s) at 298 K
if ᐃGfo values are at 25oC - 109.56 kJ/mol for AgCl(s) and - 130.79 kJ/mol for (H+ + Cl-) (aq)
Pt | H2 (1atm) | HCl (0.01 M) | AgCl(s) | Ag(s) at 298 K
if ᐃGfo values are at 25oC - 109.56 kJ/mol for AgCl(s) and - 130.79 kJ/mol for (H+ + Cl-) (aq)
Options
A.456 mV
B.654 mV
C.546 mV
D.None of these
Solution
ᐃGocell reaction = 2 (-130.79) - 2 (- 109.56) = - 42.46 kJ/mole
(for H2 + 2AgCl → 2Ag + 2H+ + 2Cl-)
∴ Eocell =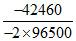 = + 0.220 V
= + 0.220 V
Now Ecell = + 0.220 +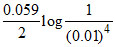 = 0.456 V = 456 mV.
= 0.456 V = 456 mV.
(for H2 + 2AgCl → 2Ag + 2H+ + 2Cl-)
∴ Eocell =
Now Ecell = + 0.220 +
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
In a cell that utilises the reaction Zn(s) + 2H+(aq) → Zn2+(aq) + H2(g) addition of H2SO4 to cathode compartment, ...In the given figure the electrolytic cell contains 1 L of an aqueous 1 M Copper (II) sulphate solution. If 0.4 mole of e...Which is/are correct among the following ?Given, the half cell emf′s EoCu+2|Cu = 0.337, EoCu+1|Cu = 0.521...Given below are the half-cell reactions Mn2+ + 2e- → Mn ; Eo = - 1.18 V (Mn3+ + e- → Mn2+) ; Eo = + 1.51 V T...An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to...