ElectrochemistryHard
Question
Which is/are correct among the following ?
Given, the half cell emf′s EoCu+2|Cu = 0.337, EoCu+1|Cu = 0.521
Given, the half cell emf′s EoCu+2|Cu = 0.337, EoCu+1|Cu = 0.521
Options
A.Cu+1 disproportionates
B.Cu and Cu2+ comproportionates.
C.EoCu | Cu+2 + EoCu+1 | Cu is positive
D.All of these
Solution
2Cu+1 → Cu + Cu+2
2Cu+1 + 2e → 2Cu
Cu - 2e → Cu+2
_______________
2Cu+1 → Cu+2 + Cu
_______________
∴ Eo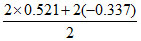 = 0.184
= 0.184
2Cu+1 + 2e → 2Cu
Cu - 2e → Cu+2
_______________
2Cu+1 → Cu+2 + Cu
_______________
∴ Eo
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Given : Hg22+ + 2e → 2Hg , Eo = 0.789 V & Hg2+ + 2e → Hg, Eo = 0.854 V, calculate the equilibrium constant f...Electrode potential data are given below.Fe3+ (aq) + e- → Fe2+ (aq); Eo = + 0.77 Al3+ (aq) + 3e- → Al (s); E...When 0.1 mol MnO42- is oxidised the quantity of electricity required to completely oxidise MnO42- to MnO4- is...Which of the following pair of metals, when coupled, will give maximum EMF for a voltaic cell?...The electric charge for electrode deposition of one gram equivalent of a substance is :...