ElectrochemistryHard
Question
In the given figure the electrolytic cell contains 1 L of an aqueous 1 M Copper (II) sulphate solution. If 0.4 mole of electrons passed through of cell, the concentration of copper ion after passage of the charge will be
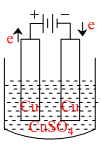

Options
A.0.4 M
B.0.8 M
C.1.0 M
D.1.2 M
Solution
Number of moles of Cu2+ discharged from anode = number of moles of Cu2+ deposited at cathode.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A hydrogen electrode placed in a buffer solution of CH3COONa and CH3COOH in the ratios of x : y and y : x has electrode ...Calculate Ka of acetic acid if its 0.05 M solution has molar conductivity of 7.814 × 10−4 Ω−1 m2 mol−1 at 25°C. Given: $...The ECell for Ag(s)|AgI (satd)||Ag+ (0.10 M)|Ag (s) is +0.413 V. What is the value of Ksp of AgI?...For a spontaneous reaction the ᐃG, equilibrium constant (K) and Eocell will be respectively...An electrochemical cell is set up as follows: Pt|H2(g) (1 atm)|0.001 M HCl||0.1 M HA|H2(g) (1 atm)|Pt EMF of this cell i...