ElectrochemistryHard
Question
At 298K the standard free energy of formation of H2O(l) is -237.20 kJ/mole while ᐃGo of its ionisation into H+ ion and hydroxyl ions is 80 kJ/mole, then the emf of the following cell at 298 K will be
H2(g,1 bar) | H+ (1M) || OH- (1M) | O2 (g, 1bar)
H2(g,1 bar) | H+ (1M) || OH- (1M) | O2 (g, 1bar)
Options
A.0.40 V
B.0.81 V
C.1.23 V
D.-0.40 V
Solution
Cell reaction Cathode : H2O(l) + 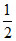 O2(g) + 2e- → 2OH-(aq.)
O2(g) + 2e- → 2OH-(aq.)
Anode : H2(g) → 2H+ (aq.) + 2e-
____________________________________
H2O(l) +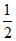 O2(g) + H2(g) → 2H+(aq.) + 2OH-(aq.)
O2(g) + H2(g) → 2H+(aq.) + 2OH-(aq.)
Also we have
H2(g) +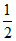 O2(g) → H2O(l) ᐃGof = - 237.2 kJ/mole
O2(g) → H2O(l) ᐃGof = - 237.2 kJ/mole
H2O(l) → H+(aq.) + OH- (aq.) ᐃGo = 80 kJ/mole
Hence for cell reaction
ᐃGo = -77.20 kJ/mole So, Eo = -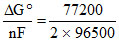 = 0.40 V
= 0.40 V
Anode : H2(g) → 2H+ (aq.) + 2e-
____________________________________
H2O(l) +
Also we have
H2(g) +
H2O(l) → H+(aq.) + OH- (aq.) ᐃGo = 80 kJ/mole
Hence for cell reaction
ᐃGo = -77.20 kJ/mole So, Eo = -
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Molar conductivities at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. ...The potential (EMF) of a cell consisting of an anode of silver in 0.10 M – AgNO3 solution and a cathode of Pt immersed i...Element A (atomic mass = 112) and element B (atomic mass = 27) form chlorides. Solutions of these chlorides are electrol...A current is passed through 500 mL of an aqueous solution of CaI2. After sometime, it is observed that 50 millimoles of ...Calculate the cell EMF in mV for Pt | H2 (1atm) | HCl (0.01 M) | AgCl(s) | Ag(s) at 298 Kif ᐃGfo values are at 25o...