ElectrochemistryHard
Question
Molar conductivities 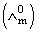 at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively.
at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. 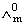 for CH3COOH will be:
for CH3COOH will be:
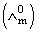 at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively.
at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. 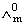 for CH3COOH will be:
for CH3COOH will be:Options
A.390.5 S cm2 mol-1
B.425.5 S cm2 mol-1
C.180.5 S cm2 mol-1
D.290.8 S cm2 mol-1
More Electrochemistry Questions
The standard electrode potentials (reduction) of Pt/Fe2+, Fe3+ and Pt/Sn4+, Sn2+ are + 0.77 V and 0.15 V respectively at...The electric charge for electrode deposition of one gram equivalent of a substance is :...In which of the following cell (s) : Ecell = Eocell ?...A cell contains two hydrogen electrodes. The negative electrode is in contact with a solution of 10−6 M hydrogen ions. T...The EMF of the cell: Hg(l)|Hg2Cl2(s), KCl sol. (1.0N)|Quinohydrone|Pt, is 0.210 V at 298 K. What is the pH of the quinoh...
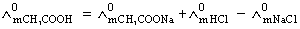 = 91.0 + 425.9 - 126.4 = 390.5
= 91.0 + 425.9 - 126.4 = 390.5