ElectrochemistryHard
Question
A current is passed through 500 mL of an aqueous solution of CaI2. After sometime, it is observed that 50 millimoles of I2 have been formed. Which of the following statements is(are) correct ?
Options
A.The number of faradays of charge passed through the solution is 0.10 F.
B.The volume of dry H2 at STP that has been formed during electrolysis is 1120 mL.
C.The pH of the solution is nearly 0.7.
D.The mass of calcium produced is 2.0 g.
Solution
CaI2(aq): Cathode Ca2+, H+
+
Anode I- OH-
×
a.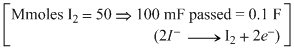
b. Also, mmoles of H2 =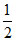 × number of mF
× number of mF
=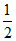 × 100 = 50 VH2 at STP
× 100 = 50 VH2 at STP
= 50 × 22.4 ml = 1120 mL
c. 100 mmoles OH- left
⇒ [OH-] =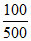 = 0.2
= 0.2
⇒ pOH = - log 0.2 = 0.7
⇒ pH = 13.3
+
Anode I- OH-
×
a.
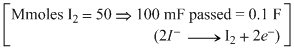
b. Also, mmoles of H2 =
=
= 50 × 22.4 ml = 1120 mL
c. 100 mmoles OH- left
⇒ [OH-] =
⇒ pOH = - log 0.2 = 0.7
⇒ pH = 13.3
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Red hot carbon will remove oxygen from the oxide AO and BO but not from MO, while B will remove oxygen from AO. The acti...The standard oxidation potentials, Eo, for the half reactions are asZn → Zn2+ + 2e- ; Eo = + 0.76 V → Fe ...Which of the following is not true about e.m.f. of a cell ?...Electrolysis of a solution of MnSO4 in aqueous sulphuric acid is a method for the preparation of MnO2 as per the reactio...A galvanic cell is composed of two hydrogen electrodes, one of which is a standard one. In which of the following soluti...