ElectrochemistryHard
Question
The specific conductivity of a saturated solution of AgCl is 3.40 × 10-6 ohm-1 cm-1 at 25oC. Ifλ Ag+ = 62.3 ohm-1 cm2 mol-1 &λCl = 67.7 ohm-1 cm2 mol-1, the solubility of AgCl at 25oC is :
Options
A. 2.6 × 10-5 M
B.2.5 × 10-13
C.25 × 10-9
D.10-6
Solution
1.53 = 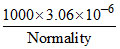
Normality = 2 × 10-3 M
Molarity =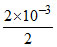 = 10-3 M
= 10-3 M
Ksp = 10-6 M
Normality = 2 × 10-3 M
Molarity =
Ksp = 10-6 M
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The resistance of a solution A is 50 Ω and that of solution B is 100 Ω, both solutions being taken in the same conductiv...Conductance measurements can be used to detect the end point of acid-base titrations. Which of the following plots corre...The reaction, H2(g) + AgCl(s) → H+ (aq) + Cl- (aq) + Ag(s) occurs in the galvanic cell :...Four colourless salt solutions are placed in separate test tubes and a strip of copper is placed in each. Which solution...Two students use same stock solution of ZnSO4 but different solutions of CuSO4. The EMF of one cell is 0.03 V higher tha...