ElectrochemistryHard
Question
Conductance measurements can be used to detect the end point of acid-base titrations. Which of the following plots correctly represent the end point of the titration of strong acid and a strong base ?
Options
A.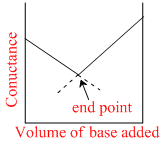

B.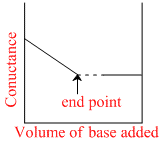

C.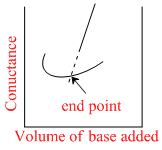

D.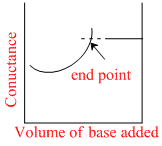

Solution
H+ + CI- + NaOH → Na+ + CI- + H2O to conductance Ist decreases since no. of ions decreases after end point it inceases.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Which is Lewis acid ?...The specific conductance of a N/10 KCl at 25oC is 0.0112 ohm-1 cm-1. The resistance of cell containing solution at the s...The molar conductivity of NH4Cl, OH− and Cl− at infinite dilution is 150, 200 and 75 Ω−1 cm2 mol−1, respectively. If the...Three faradays of electricity is passed through molten Al2O3, aqueous solutions of CuSO4 and molten NaCl. The amounts of...Given standard electrode potentials : Fe3+ + 3e- → Fe ; Eo = - 0.036 voltFe2+ + 2e- → Fe; Eo = - 0.440 volt ...