ElectrochemistryHard
Question
The ionization constant of a weak electrolyte is 25 × 10-6 while the equivalent conductance of its 0.01 M solution is 19.6 S cm2 eq-1. The equivalent conductance of the electrolyte at infinite dilution (in S cm2 eq-1 ) will be
Options
A.250
B.196
C.392
D.384
Solution
Ka = 25 × 10-6 Λeq = 19.6 Scm2 eq-1, C = 0.01
Ka = 0.01 × α2 ⇒ α =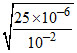 = 5 × 10-2
= 5 × 10-2
α = 5 × 10-2 =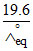 ⇒
⇒ 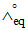 =
= 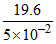 = 392 Scm2 eq-1.
= 392 Scm2 eq-1.
Ka = 0.01 × α2 ⇒ α =
α = 5 × 10-2 =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The solubility product of silver iodide is 8.3 × 10-17 and the standard reduction potential of Ag, Ag+ electrode is...When a lead storage battery is discharged...Using the information in the preceding problem, calculate the solubility product of AgI in water at 25oC [Eo(Ag+, Ag) = ...Molar conductance of BaCl2, H2SO4 and HCl at infinite dilutions are x1, x2 and x3 respectively.Equivalent conductance of...The cell Pt (H2) (1 atm) | (pH = ?) || (a = 1) | AgI(s), Ag has emf, E298K = 0. The standard electrode potential for the...