ElectrochemistryHard
Question
Using the information in the preceding problem, calculate the solubility product of AgI in water at 25oC [Eo(Ag+, Ag) = + 0.799 volt]
Options
A.1.97 × 10-17
B.8.43 × 10-17
C.1.79 × 10-17
D.9.17 × 10-17
Solution
EoAg|AgI|I- = EoAg+|Ag - 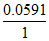 log
log 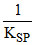
- 0.151 = 0.799 -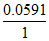 log
log 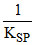
0.0591 log Ksp = - 0.151 -0.799
log Ksp = - 16.074
Ksp = 8.43 × 10-17
- 0.151 = 0.799 -
0.0591 log Ksp = - 0.151 -0.799
log Ksp = - 16.074
Ksp = 8.43 × 10-17
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A hydrogen electrode placed in a buffer solution of CH3COONa and CH3COOH in the ratio’s x:y and y:x has electrode potent...Select correct statement:...When molten ICI3 is electrolysed using platinum electrodes...The current required to produce oxygen at the rate of 2.8 ml (0°C, 1 atm) per second during electrolysis of acidulated w...The standard reduction potentials at 25°C of Li+|Li, Ba2+|Ba, Na+|Na and Mg2+|Mg are −3.05, −2.73, −2.71 and −2.37 V, re...