ElectrochemistryHard
Question
Molar conductance of BaCl2, H2SO4 and HCl at infinite dilutions are x1, x2 and x3 respectively.Equivalent conductance of BaSO4 at at infinite dilution will be:
Options
A.(x1 + x2 - x3)/2
B.x1 + x2 - 2x3
C.(x1 - x2 - x3)/2
D.(x1 + x2 - 2x3)/2
Solution
λm∞(BaSO4) =λBa2+∞ + λSO42+∞
= λm∞(BaCl2) + λm∞(H2SO4)- - 2λ(HCl)∞
= x1 + x2 - 2x3 ; λeo =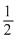 λm∞(BaSO4)
λm∞(BaSO4)
= λm∞(BaCl2) + λm∞(H2SO4)- - 2λ(HCl)∞
= x1 + x2 - 2x3 ; λeo =
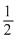 λm∞(BaSO4)
λm∞(BaSO4)Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
For the reduction of silver ions with copper metal, the standard cell potential was found to be +0.46 V at 25oC. The val...A student made the following observations in the laboratory:(I) Clean copper metal did not react with 1 M – Pb(NO3)2 sol...IV CH2 = NHAmong these compounds, the correct order of C - N bond lengths is :...Sodium amalgam is prepared by electrolysis of aqueous NaCl using 10 g mercury as cathode. How many Faraday of electricit...Amount of oxalic acid present in a solution can be determined by its titration with KMnO4 solution in the presence of H2...