ElectrochemistryHard
Question
The solubility product of silver iodide is 8.3 × 10-17 and the standard reduction potential of Ag, Ag+ electrode is + 0.8 volts at 25oC. The standard reduction potential of Ag,Agl/l- electrode from these data is
Options
A.- 0.30 V
B.+ 0.15 V
C.+ 0.10 V
D.- 0.15 V
Solution
EAg/AgI/I- = 0.8 - 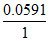 log
log 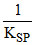
= 0.8 + 0.0591 × log 8.3 × 10-17
= - 0.15 Volt
or
EAg/AgI/I- = EoAg/Ag+ +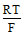 . ln
. ln 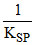
= - 0.8 -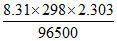 log ksp
log ksp
= - 0.8 - 0.0591. log 8.3 × 10-17
= - 0.8 + 0.095 = 0.15 V.
= 0.8 + 0.0591 × log 8.3 × 10-17
= - 0.15 Volt
or
EAg/AgI/I- = EoAg/Ag+ +
= - 0.8 -
= - 0.8 - 0.0591. log 8.3 × 10-17
= - 0.8 + 0.095 = 0.15 V.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A cell whose resistance, when filled with 0.1 M – KCl is 200 Ω, is measured to be 6400 Ω, when filled with 0.003 M – NaC...Element A (atomic mass = 112) and element B (atomic mass = 27) form chlorides. Solutions of these chlorides are electrol...Assuming that copper contains only iron, silver and gold as impurities. After passage of 12.4 A for 4825 s, the mass of ...The oxidation number of sulphur in H2S2O7 is :...The number of Faradays required to produce 1 g-atom of Mg from MgCl2 is...