ElectrochemistryHard
Question
The cell Pt (H2) (1 atm) | (pH = ?) || (a = 1) | AgI(s), Ag has emf, E298K = 0. The standard electrode potential for the reaction AgI + e- → Ag + IQ is - 0.151 volt. Calculate the pH value.
Options
A.3.37
B.5.26
C.2.56
D.4.62
Solution
0 = (- 0.151 - 0) - 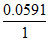 log [H+].
log [H+].
0.0591 × log[H+] = - 0.151
pH =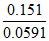 = 2.56
= 2.56
0.0591 × log[H+] = - 0.151
pH =
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
...The gas x at 1 atm is bubbed through a solution containig a mixure of 1 My- and Mz- at 25oC, If the order of reduction p...In the electrolysis of acidified AgNO3 solution using Pt-electrodes, the anode reaction is...When molten ICI3 is electrolysed using platinum electrodes...The molar conductance of a strong electrolyte at infinite dilution...