ElectrochemistryHard
Question
Three faradays of electricity was passed through an aqueous solution of iron (II) bromide. The mass of iron metal (at. mass 56) deposited at the cathode is -
Options
A.56 g
B.84 g
C.112 g
D.168 g
Solution
Mole of Fe deposited = 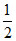 × 3 = 1.5 mole
× 3 = 1.5 mole
WFe = 1.5 × 56 = 84 gm.
WFe = 1.5 × 56 = 84 gm.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Which of the following curve represents the variation of λM with √C for AgNO3 ?...Choose the correct statement(s)....What is cell entropy change of the following cell ?Pt(s) | H2 (g) | CH3COOH, HCl || KCl (aq) | Hg2Cl2 (s) | Hg P = 1 atm...What is the pH of 0.01 m glycine solution ? for glycine, Ka1 = 4.5 × 10-3 and Ka2 = 1.7 × 10-10 at 298K ?...The conductivity of a solution of AgCl at 298 K is found to be 1.382 × 10-6 W-1 cm-1. The ionic conductance of Ag+ ...