ElectrochemistryHard
Question
What is cell entropy change of the following cell ?
Pt(s) | H2 (g) | CH3COOH, HCl || KCl (aq) | Hg2Cl2 (s) | Hg
P = 1 atm 0.1M 0.1M
Emf of the cell is found to be 0.045 V at 298 K and temperature coefficient is 3.4 × 10-4 V K-1
Given Ka(CH3COOH) = 10-5 M
Pt(s) | H2 (g) | CH3COOH, HCl || KCl (aq) | Hg2Cl2 (s) | Hg
P = 1 atm 0.1M 0.1M
Emf of the cell is found to be 0.045 V at 298 K and temperature coefficient is 3.4 × 10-4 V K-1
Given Ka(CH3COOH) = 10-5 M
Options
A.60
B.65.2
C.69.2
D.63.5
Solution
Considering the cell reaction
H2 + 2e → 2H+
Hg22+ + 2e → 2Hg (l) + 2Cl- (aq)
ᐃS = nF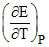 = 2 × 96500 × 3.4 × 10-4 = 65.223 J/K/mole
= 2 × 96500 × 3.4 × 10-4 = 65.223 J/K/mole
H2 + 2e → 2H+
Hg22+ + 2e → 2Hg (l) + 2Cl- (aq)
ᐃS = nF
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Which of the following statement(s) differentiate between electrochemical cell and electrolytic cell?...Pt (where p1 and p2 are pressures) cell reaction will be spontaneous if :...The standard reduction potential of oxygen in acidic solution is +1.23 V. What is the standard reduction potential of ox...Which statement is correct....In which of the following cells, EMF is greater than E-cell ?...