ElectrochemistryHard
Question
The conductivity of a solution of AgCl at 298 K is found to be 1.382 × 10-6 W-1 cm-1. The ionic conductance of Ag+ and Cl- at infinite dilution are 61.9 W-1 cm2 mol-1 and 76.3 W-1 cm2 mol-1, respecitvley. The solubility of AgCl is
Options
A.1.4 × 10-5 mol L-1
B.1 × 10-2 mol L-1
C.1 × 10-5 mol L-1
D.1.9 × 10-5 mol L-1
Solution
K = 1.382 × 10-6 s cm-1
ΛAgCl = 61.9 + 76.3 = 138.2 =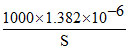
S = 10-5 M.
ΛAgCl = 61.9 + 76.3 = 138.2 =
S = 10-5 M.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
For the process: Cu2+ + 2e− → Cu; log[Cu2+] vs. Ered graph is shown in the figure, where OA = 0.34 V. The electrode pote...An electrolytic cell contains a solution of Ag2SO4 and platinum electrodes. A current is passed until 1.6 g of O2 has be...The standard reduction potential data at 25oC is given belowEo (Fe3+, Fe2+) = + 0.77 V ;Eo (Fe2+, Fe) = - 0.44 V ;Eo (Cu...The following galvanic cell: Zn|Zn(NO3)2 (100 ml, 1 M)||Cu(NO3)2 (100 ml, 1 M)|Cu was operated as an electrolytic cell a...Beryllium is placed above magnesium in the group II. Beryllium dust, therefore, when added to MgCl2 solution will...