ElectrochemistryHard
Question
What is the pH of 0.01 m glycine solution ? for glycine, Ka1 = 4.5 × 10-3 and Ka2 = 1.7 × 10-10 at 298K ?
Options
A.3.0
B.10.0
C.6.1
D.7.1
Solution
For glycine solution ;
K = Ka1 × Ka2
= 4.5 × 10-13 × 1.7 × 10-10
= 7.65 × 10-13+] =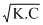
=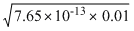 = 8.7 × 10-8
= 8.7 × 10-8
Hence, pH = - log[H+]
= - log[8.7 × 10-8]
= - (log10-8 + log8.7)
8 - 0.93 = 7.07 ≈ 7.1
K = Ka1 × Ka2
= 4.5 × 10-13 × 1.7 × 10-10
= 7.65 × 10-13+] =
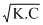
=
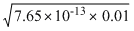 = 8.7 × 10-8
= 8.7 × 10-8Hence, pH = - log[H+]
= - log[8.7 × 10-8]
= - (log10-8 + log8.7)
8 - 0.93 = 7.07 ≈ 7.1
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Zinc granules are added in excess to a 500 ml of 1.0 M nickel nitrate solution at 25°C until the equilibrium is reached....What is the emf at 25oC for the cell, Ag Pt The standard reduction potentials for the half-reactions AgBr + e- → A...A depolarizer used in dry cell batteries is...The molar conductance of a strong electrolyte at infinite dilution...The ECell for Ag(s)|AgI (satd)||Ag+ (0.10 M)|Ag (s) is +0.413 V. What is the value of Ksp of AgI?...