ElectrochemistryHard
Question
Choose the correct statement(s).
Options
A.At the anode, the species having minimum reduction potential is formed from the oxidation of corresponding oxidizable species.
B.In highly alkaline medium, the anodic process during the electrolytic process is
4OH- → O2 + 2H2O + 4e-.
4OH- → O2 + 2H2O + 4e-.
C.The standard potential of Cl- |AgCl| Ag half-cell is related to that of Ag+ | Ag through the expression
EoAg+|Ag = EoCl-|AgCl|Ag +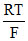 ln Ksp(AgCl)
ln Ksp(AgCl)
EoAg+|Ag = EoCl-|AgCl|Ag +
D.Compounds of active metals (Zn, Na, Mg) are reducible by H2 whereas those of noble metals (Cu, Ag, Au) are not reducible.
Solution
In highly alkaline medium OH- ion get oxidise in preference to H2O
4OH- → O2 + 2H2O + 4e-
EoCl-/AgCl/Ag = EoAg+/Ag -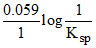
EoRed of active metal is lower than EoRed of hydrogen so not reduced by hydrogen.
4OH- → O2 + 2H2O + 4e-
EoCl-/AgCl/Ag = EoAg+/Ag -
EoRed of active metal is lower than EoRed of hydrogen so not reduced by hydrogen.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Pt (where p1 and p2 are pressures) cell reaction will be spontaneous if :...The standard reduction potential of oxygen in acidic solution is +1.23 V. What is the standard reduction potential of ox...The efficiency of an hypothetical cell is about 84% which involves the following reaction :A (s) + B2+ (aq) → A2+ ...In a cell that utilises the reaction Zn(s) + 2H+(aq) → Zn2+(aq) + H2(g) addition of H2SO4 to cathode compartment, ...At 25°C, the solubility product of CuCl is 2.0 × 10−7 and $E_{Cl^{-}|CuCl|Cu}^{o}$ is 0.128 V. The value of $E_{Cu^{+}|C...