SolutionHard
Question
The vapour pressure of a pure liquid A is 40 mmHg at 310 K. The vapour pressure of this liquid in a solution with liquid B is 32 mmHg. Mole fraction of A in the solution, if it obeys Raoult′s law is :
Options
A.0.8
B.0.5
C.0.2
D.0.4
Solution
pA = XA poA
32 = XA 40
∴ XA =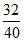 = 0.8.
= 0.8.
32 = XA 40
∴ XA =
Create a free account to view solution
View Solution FreeMore Solution Questions
A vessel has nitrogen gas and water vapours in equilibrium with liquid water at a total pressure of 1 atm. The partial p...The molarity of H2SO4 solution, which has a density 1.84g/cc. at 35oC and contains 98% by weight, is...The freezing point depression constant for water is 1.86oC. If 5.00 g Na2SO4 is dissolved in 45.0 g H2O, the freezing po...Which of the following combinations are correct for a binary solution, in which the solute as well as solvent are liquid...The fundamental cause of ᐃT (depression is) :...