SolutionHard
Question
A vessel has nitrogen gas and water vapours in equilibrium with liquid water at a total pressure of 1 atm. The partial pressure of water vapours is 0.3 atm. The volume of this vessel is reduced to one third of the original volume, at the same temperature, then total pressure of the system is :(Neglect volume occupied by liquid water)
Options
A.3.0 atm
B.1 atm
C.3.33 atm
D.2.4 atm
Solution
PN2 + PH2O(v) = 1 atm, ∵ PH2O = 0.3 atm
∴ PN2 = 0.7 atm
Now new pressure of N2 in another vessel of volume V/3 at same T is given by :
PN2 ×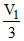 = 0.7 0 × V1
= 0.7 0 × V1
∴ PN2 = 2.1 atm
Since aqueous tension remains constant, and thus total pressure in new vessel.
= PN2 + PH2O = 2.1 + 0.3 = 2.4 atm.
∴ PN2 = 0.7 atm
Now new pressure of N2 in another vessel of volume V/3 at same T is given by :
PN2 ×
∴ PN2 = 2.1 atm
Since aqueous tension remains constant, and thus total pressure in new vessel.
= PN2 + PH2O = 2.1 + 0.3 = 2.4 atm.
Create a free account to view solution
View Solution FreeMore Solution Questions
What is the OH- concentration of an aqueous solution of a substance whose pH = 3.2 :-...The pH vlues of aqueous solutions of which of the following compounds does not change on dilution ?...The concentration of pollutant in ppm (w/w), that has been measured at 450 mg per 150 kg of sample is :...Consider the following reactions.$${PbCl}_{2} + K_{2}{CrO}_{4} \rightarrow \text{ }A + 2KCl $$(Hot solution)$${A + NaOH ...Heptane and octane form ideal solution. At 373 K, the vapour pressures of the pure liquids are 106 kPa and 46 kPa, respe...