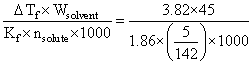SolutionHard
Question
The freezing point depression constant for water is 1.86oC. If 5.00 g Na2SO4 is dissolved in 45.0 g H2O, the freezing point is changed by 3.82oC. Calculate the van′t Hoff factor for Na2SO4.
Options
A.2.63
B.3.11
C.0.381
D.2.05
More Solution Questions
What is the OH- concentration of an aqueous solution of a substance whose pH = 3.2 :-...Which is/are correct statement(s) ?...In which case, van′t Hoff factor i remains unchanged ? [Assume common complexes of these ions]...Consider the following four liquids :1. Water : highly polar ; H-bonding2. Hexanol : slightly polar ; some H-bonding3. C...Barium ions, CN- and Co2+ form an ionic complex. If that complex is supposed to be 75% ionised in water with vant Hoff f...