SolutionHard
Question
The molarity of H2SO4 solution, which has a density 1.84g/cc. at 35oC and contains 98% by weight, is
Options
A.1.84 M
B.18.4 M
C.20.6 M
D.24.5 M
Solution
100 gm solution contains 98 gm H2SO4 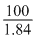 cc. contains 98 gm H2SO4
cc. contains 98 gm H2SO4
= 100c.c solution contains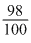 × 1.84 × 1000 gm H2SO4
× 1.84 × 1000 gm H2SO4
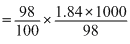 moles of H2SO4
moles of H2SO4
= 18.4 M.
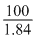 cc. contains 98 gm H2SO4
cc. contains 98 gm H2SO4 = 100c.c solution contains
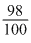 × 1.84 × 1000 gm H2SO4
× 1.84 × 1000 gm H2SO4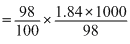 moles of H2SO4
moles of H2SO4 = 18.4 M.
Create a free account to view solution
View Solution FreeMore Solution Questions
Which facts are true when we use van′t Hoff equation PV = CST for osmotic pressure P of dilute solutions ?...Pressure cooker reduces cooking time because...Which of the following solution can act as buffer :-...Mixture of volatile components A and B has total vapour pressure (in Torr) p = 254 - 119 xA where xA is mole fraction of...Y gm of non-volatile organic substance of molecular mass M is dissolved in 250 gm benzene. Molal elevation constant of b...