Solid StateHard
Question
If the anions (A) form hexagonal closest packing and cations (C) occupy only 2/3 octahedral voids in it, then the general formula of the compound is
Options
A.CA
B.CA2
C.C2A3
D.C3A2
Solution
No. of A atoms = 6.
No. of C atoms = 6 ×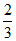 = 4.
= 4.
∴ Formula = C4A6 or C2A3.
No. of C atoms = 6 ×
∴ Formula = C4A6 or C2A3.
Create a free account to view solution
View Solution FreeMore Solid State Questions
An ionic compound has a unit cell consisting of A ions at the corners of a cube and B ions on the centres of the faces o...Which of the following statements is/are false....The densities of ice and water at 0°C and 1 bar are 0.96 and 0.99 g/cm3, respectively. If the percentage of occupied spa...NaCl is doped with 2 × 10–3 mole% SrCl2, the concentration of cation vacancies is (NA = 6 × 1023)...A solid element (monoatomic) exists in cubic crystal. If its atomic radius is 1.0 Å and the ratio of packing fraction an...