Solid StateHard
Question
An ionic compound has a unit cell consisting of A ions at the corners of a cube and B ions on the centres of the faces of the cube. The empirical formula for this compound would be
Options
A.AB
B.A2B
C.AB3
D.A3B
Solution
A = 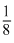 × 8 = 1
× 8 = 1
(Corner)
B =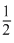 × 6 = 3
× 6 = 3
(Face centre)
∴ AB3
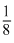 × 8 = 1
× 8 = 1(Corner)
B =
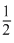 × 6 = 3
× 6 = 3(Face centre)
∴ AB3
Create a free account to view solution
View Solution FreeMore Solid State Questions
How many unit cells are present in a cubeshaped ideal crystal of NaCl of mass 1.00 g? [Atomic masses: Na = 23, Cl = 35.5...Na and Mg crystallize in BCC and FCC type crystals respectively, then the number of atoms of Na and Mg present in the un...The density of KCl is 1.9893 g-cm-3 and the length of a side of unit cell is 6.29082 A0. The value of Avogadro′s n...In a sodium chloride structure, the percentage of the octahedral voids occupied by cations is :...Following three planes( P1, P2, P3) in an FCC unit cell are shown: Consider the following statements and choose the corr...