Atomic StructureHard
Question
1st excitation potential for the H-like (hypothetical) sample is 24 V. Then :
Options
A.Ionisation energy of the sample is 36 eV
B.Ionisation energy of the sample is 32 eV
C.Binding energy of 3rd excited state is 2 eV
D.2nd excitation potential of the sample is 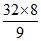 V
V
Solution
1st excitation potential = 10.2 Z2 = 24 V ∴ Z2 = 24/10.2
∴ IE = 13.6 Z2 =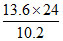 = 32 eV.
= 32 eV.
Binding energy of 3rd excited state = 0.85 Z2 =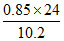 = 2eV.
= 2eV.
2nd excitation potential of sample = 12.09 Z2 =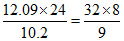 V.
V.
∴ IE = 13.6 Z2 =
Binding energy of 3rd excited state = 0.85 Z2 =
2nd excitation potential of sample = 12.09 Z2 =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Photon having energy equivalent to the binding energy of 4th state of He+ atom is used to eject an electron from the met...A gas absorbs a photon of 355 nm and emits at two wavelengths. If one of the emissions is at 680 nm, the other is at :...Ionisation energy of He+ is 19.6 × 10-18 J atom-1. The energy of the first stationary state (n = 1) of Li2+ is...The group in which all the elements do not have same number of electrons in their valence shell is :-...In an atom, an electron is moving with a speed of 600m/s with an accuracy of 0.005%. Certainity with which the position ...