Atomic StructureHard
Question
Photon having energy equivalent to the binding energy of 4th state of He+ atom is used to eject an electron from the metal surface of work function 1.4 eV. If electrons are further accelerated through the potential difference of 4V then the minimum value of De-broglie wavelength associated with the electron is :
Options
A.1.1 (Ao)
B.5 (Ao)
C.9.15 (Ao)
D.11 (Ao)
Solution
Total energy = 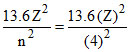 = 3.4 eV
= 3.4 eV
Now K.E. = 3.4 - 1.4 = 2 eV
Now, Total energy = 2 + 4 = 6 eV i.e. potential = 6 V
For electron λ =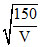 so λ = 5 (Ao).
so λ = 5 (Ao).
Now K.E. = 3.4 - 1.4 = 2 eV
Now, Total energy = 2 + 4 = 6 eV i.e. potential = 6 V
For electron λ =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The wavelength of radiation emitted out in the transition n = 4 to n = 1 in Li2+ ion is...The potential energy of the electron present in the ground state of Be3+ ion is represented by:...Because of lanthanoid contraction, which of the following pairs of elements have nearly same atomic radii ? (Nubmers in ...In Bohr’s model of the hydrogen atom, let r, v and E represent the orbit radius, speed of an electron and the total ener...What would be the approximate quantum number (n) for a circular orbit of hydrogen, 1 × 10–5 cm in diameter?...