Atomic StructureHard
Question
In an atom, an electron is moving with a speed of 600m/s with an accuracy of 0.005%. Certainity with which the position of the electron can be located is (h = 6.6 × 10-34 kg m2s-1, mass of electron, em = 9.1 × 10-34 kg)
Options
A.1.52 × 10-4 M
B.5.10 × 10-3 M
C.9.92 × 10-3 M
D.3.84 × 10-3 M
Solution
ᐃx.m ᐃv = 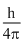
ᐃx =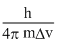
ᐃv = 600 ×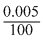 = 0.03
= 0.03
⇒ ᐃx =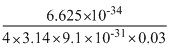 =1.92 × 10-3 m
=1.92 × 10-3 m
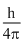
ᐃx =
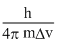
ᐃv = 600 ×
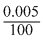 = 0.03
= 0.03⇒ ᐃx =
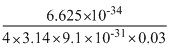 =1.92 × 10-3 m
=1.92 × 10-3 mCreate a free account to view solution
View Solution FreeMore Atomic Structure Questions
A photon of 2.55 eV is emitted out by an electronic transition in hydrogen atom. The change in de Broglie wavelength of ...A bulb emits light of wavelength 1987 8 7.nm. The bulb is rated as 200 W and 14% of the energy is emitted as light. How ...If there are three possible values (–1/2, 0, +1/2) for the spin quantum number, then the maximum capacity of second orbi...Bohr′s model can explain :...In two hydrogen atoms A and B, the electrons are revolving around the nucleus in circular orbits of radius r and 4r, res...