Atomic StructureHard
Question
Ionisation energy of He+ is 19.6 × 10-18 J atom-1. The energy of the first stationary state (n = 1) of Li2+ is
Options
A.4.41 × 10-16 J atom-1
B.- 4.41 × 10-17 J atom-1
C.- 2.2 × 10-15 J atom-1
D.8.82 × 10-17 J atom-1
Solution
IEHe+ = 13.6 Z2He+ 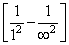 = 13.6 Z2He+ where (Z2He+ = 2)
= 13.6 Z2He+ where (Z2He+ = 2)
Hence, 13.6 × Z2He+ = 19.6 × 10-18 J atom-1
(E1)Li++ = - 13.6 Z2Li+2 x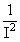 = 13.6 Z2He+ ×
= 13.6 Z2He+ ×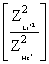 ×19.6 × 10-18 ×
×19.6 × 10-18 × 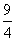
= 4.41 × 10-17 J atom-1
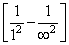 = 13.6 Z2He+ where (Z2He+ = 2)
= 13.6 Z2He+ where (Z2He+ = 2)Hence, 13.6 × Z2He+ = 19.6 × 10-18 J atom-1
(E1)Li++ = - 13.6 Z2Li+2 x
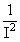 = 13.6 Z2He+ ×
= 13.6 Z2He+ × ×19.6 × 10-18 ×
×19.6 × 10-18 × 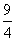
= 4.41 × 10-17 J atom-1
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In aqueous solution the ionization constants for carbonic acid are K1 = 4.2 × 10-7 and K2 = 4.8 × 10-11 Select...Bohr′s theory is not applicable to...If two py - orbital from two atoms are forming π - bond, then predict their approach axis-...n-Butane (C4H10) is produced by monobromination of C2H6 followed by Wurtz reaction. Calculate the volume of ethane at ST...The difference in angular momentum associated with the electron in two successive orbits of hydrogen atom in :-...