Atomic StructureHard
Question
A gas absorbs a photon of 355 nm and emits at two wavelengths. If one of the emissions is at 680 nm, the other is at :
Options
A.325 nm
B.743 nm
C.518 nm
D.1035 nm
More Atomic Structure Questions
Wrong statement about [Ni(CO)4] is :-...Which of the the following has maximum energy ?...Which transition in Li2+ would have the same wavelength as the 2 → 4 transition in He+ ion ?...How long (approximate) should water be electrolysed by passing through 100 amperes current so that the oxygen released...Simultaneous determination of exact position and momentum of an electron is :-...
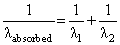
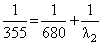
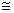 nm
nm