Chemical Kinetics and Nuclear ChemistryHard
Question
The rate of a chemical reaction doubles for every 10oC rise of temperature. If the temperature is raised by 50oC, the rate of the reaction increases by about :
Options
A.24 times
B.32 times
C.64 times
D.10 times
Solution
Temperature coefficient μ = 2.
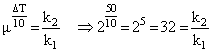
Therefore 32 k1 = k2
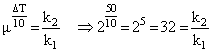
Therefore 32 k1 = k2
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
In the reaction : P + Q →R + S the time taken for 75% reaction of P is twice the time taken for 50% reaction of P. ...When initial concentration of a reactant is doubled in a reaction, its half-life period is not affected. The order of th...Graph between log k and 1/T (k is rate constant in s-1 and T is the temperature in K) is a straight line. As shown in fi...Consider the following complex ions P, Q and R ,P = [FeF6]3- , Q = [V(H2O)6]2+ and R = [Fe(H2O)6]2+The correct order of ...95Am241 and 90Th234 belong respectively to :...