AIPMT | 2015Chemical Kinetics and Nuclear ChemistryHard
Question
When initial concentration of a reactant is doubled in a reaction, its half-life period is not affected. The order of the reaction is :-
Options
A.First
B.Second
C.More than zero but less than first
D.Zero
Solution
T1/2 = 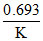 for first order t1/2 is independent of concentration
for first order t1/2 is independent of concentration
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the reaction A2(g) → B(g) + ½C(g), pressure of the system increases from 100 to 120 mm in 5 minutes. The average rat...What is ᐃH for the reaction A + B → C, where the mechanism involves several kinetic steps....For the first-order reaction (C = C0e-k1t) and Tav = K1-1. After two average lives concentration of the reactant is redu...For irreversible elementary reactions in parallel $A\overset{\quad K_{1}\quad}{\rightarrow}R\text{ and }A\overset{\quad ...First order reaction 2N2O(g) → 2N2g + O2(g)has a rate constant of 1.3 × 10-11 S-1 at 270C and 4.5 × 10-1...