Chemical Kinetics and Nuclear ChemistryHard
Question
In the reaction :
P + Q →R + S
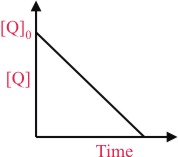
the time taken for 75% reaction of P is twice the time taken for 50% reaction of P. The concentrationof Q varies with reaction time as shown in the figure. The overall order of the reaction is -
Options
A.2
B.3
C.0
D.1
Solution
For reactant ′P′
Since t75% = 2 t50%
therefore order with respect to ′P′ is ′one′
For reactant ′Q′
linear variation of concentration of ′Q′ with time suggest that order with respect to ′Q′ is zero, therefore
overall order will be = 1 + 0 = 1
Correct Option (D)
(Assuming conc. of other reactant to be very large)
Since t75% = 2 t50%
therefore order with respect to ′P′ is ′one′
For reactant ′Q′
linear variation of concentration of ′Q′ with time suggest that order with respect to ′Q′ is zero, therefore
overall order will be = 1 + 0 = 1
Correct Option (D)
(Assuming conc. of other reactant to be very large)
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For an endothermic reaction where áƒH represents the enthalpy of the reaction in kJ/ mol, the minimum value for the ene...Incorrect order of stablility is :...Number of natural life times (Tav) required for a first-order reaction to achieve 99.9% level of completion is :...Penetrating power of α-particle is :...A certain zero order reaction has k = 0.025 M s-1 for the disappearance of A. What will be the concentration of A after ...