Chemical Kinetics and Nuclear ChemistryHard
Question
Consider the following complex ions P, Q and R ,
P = [FeF6]3- , Q = [V(H2O)6]2+ and R = [Fe(H2O)6]2+
The correct order of the complex ions, according to their spin-only magnetic moment values (in B.M.)is -
P = [FeF6]3- , Q = [V(H2O)6]2+ and R = [Fe(H2O)6]2+
The correct order of the complex ions, according to their spin-only magnetic moment values (in B.M.)is -
Options
A.R < Q < P
B.Q < R < P
C.R < P < Q
D.Q < P < R
Solution
spin only magnetic moment 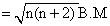
n → number of unpaired electrons
P : [FeF6]3-
F- → (weak field ligand)
ᐃ0 < P
Fe3+ ; [Ar] 3d5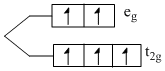
number of unpaired electron = 5
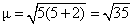
Q : [V(H2O)6]2+
H2O → (weak field ligand)
ᐃ0 < P
V2+ → [Ar]3d3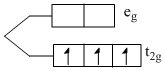
number of unpaired electron = 3
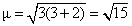
R : [Fe(H2O)6]2+
H2O → (weak field ligand)
ᐃ0 < P
Fe2+ ; [Ar] 3d6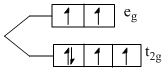
number of unpaired electron = 4
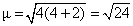
Hence, Ans (B) : Q < R < P
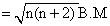
n → number of unpaired electrons
P : [FeF6]3-
F- → (weak field ligand)
ᐃ0 < P
Fe3+ ; [Ar] 3d5

number of unpaired electron = 5
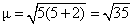
Q : [V(H2O)6]2+
H2O → (weak field ligand)
ᐃ0 < P
V2+ → [Ar]3d3

number of unpaired electron = 3
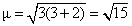
R : [Fe(H2O)6]2+
H2O → (weak field ligand)
ᐃ0 < P
Fe2+ ; [Ar] 3d6

number of unpaired electron = 4
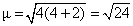
Hence, Ans (B) : Q < R < P
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
At 370 K a gaseous reaction X → 2Y + Z is found to be first order. Starting the reaction from pure X the total pre...A reaction involving two different reactants can never be...First order reaction 2N2O(g) → 2N2g + O2(g)has a rate constant of 1.3 × 10-11 S-1 at 270C and 4.5 × 10-1...The colloid is :...The composition of N2O5 is a first order reaction represented by: N2O5 → N2O4 + 1/2O2.After 20 min the volume of O...