Atomic StructureHard
Question
A nuceus 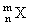 emits one α particle and two β- particles. The resulting nucleus is
emits one α particle and two β- particles. The resulting nucleus is
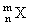 emits one α particle and two β- particles. The resulting nucleus is
emits one α particle and two β- particles. The resulting nucleus is Options
A.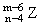
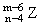
B.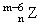
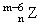
C.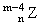
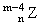
D.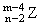
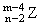
Solution
When an alpha particle 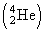 is emitted, the mass number and the atomic number of the daugther nucleus decreases by four and two respectively. When a beta particle (β-) is emitted, the atomic number of the daughter nucleus increases by one but the mass number remains the same.
is emitted, the mass number and the atomic number of the daugther nucleus decreases by four and two respectively. When a beta particle (β-) is emitted, the atomic number of the daughter nucleus increases by one but the mass number remains the same.
∴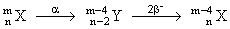
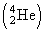 is emitted, the mass number and the atomic number of the daugther nucleus decreases by four and two respectively. When a beta particle (β-) is emitted, the atomic number of the daughter nucleus increases by one but the mass number remains the same.
is emitted, the mass number and the atomic number of the daugther nucleus decreases by four and two respectively. When a beta particle (β-) is emitted, the atomic number of the daughter nucleus increases by one but the mass number remains the same. ∴
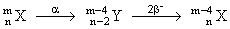
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The activity of a radioactive sample is 1.6curie and its half life is 2.5days . Them activity after 10 days will be :...A star initially has 104 deutetron. It produces energy via the processes 1H2 + 1H2 → 1H3 + p and 1H2 + 1H3 →...The Kα X-ray emission line of tungsten occurs at λ = 0.021 nm. The energy difference between K and L levels in...Two cylinders A and B fitted with pistons contain equal amounts of an ideal diatomic gas at 300 K. The piston of A is fr...Suppose an electron is attracted towards the origin by a force k/r where ′k′ is a constant and ′r̸...