Atomic StructureHard
Question
The Kα X-ray emission line of tungsten occurs at λ = 0.021 nm. The energy difference between K and L levels in this atoms is about
Options
A.0.51 MeV
B.1.2 MeV
C.59 keV
D.13.6 eV
Solution
λKα = 0.021 amu = 0.21
Since, λKα corresponding to the transition of an electron from L-shell to K-shell,
therefore
EL EK = (in eV) =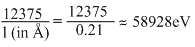
or ᐃE = 59 keV.
Since, λKα corresponding to the transition of an electron from L-shell to K-shell,
therefore
EL EK = (in eV) =
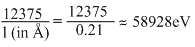
or ᐃE = 59 keV.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
When 3Li7 nuclei are bombarded by protons, and the resultant nuclei are 4Be8, the emitted particles will be...For an electron in the second orbit of hydrogen, the moment of momentum as per Bohr′s model is...A proton is about 1840 times heavier than an electron when it is accelerated by a potential difference of 1 kV, its kine...The kinetic in the potential different of 100V,...When an electron-positron pair annihilates, the energy released is about...