Atomic StructureHard
Question
A star initially has 104 deutetron. It produces energy via the processes
1H2 + 1H2 → 1H3 + p and 1H2 + 1H3 → 2He4 + n.
If the average power radiated by the star is 1016 W, the deuteron supply of the star is exhausted in a time of the order of
The mass of the nuclei are follows
M(H2) = 2.014 amu; M(n) = 1.008amu;
M(p) = 1.007 amu; M(He4) = 4.001 amu.
1H2 + 1H2 → 1H3 + p and 1H2 + 1H3 → 2He4 + n.
If the average power radiated by the star is 1016 W, the deuteron supply of the star is exhausted in a time of the order of
The mass of the nuclei are follows
M(H2) = 2.014 amu; M(n) = 1.008amu;
M(p) = 1.007 amu; M(He4) = 4.001 amu.
Options
A.106s
B.108s
C.1012s
D.1016s
Solution
The given reactions are :
1H2 + 1H2 → 1H3 + p
1H2 + 1H3 → 2He4 + n
3 1H2 → 2He4 + n + p
Mass defect
ᐃm = (3 × 2.014 - 4.001 - 1.007 - 1.008) amu
= 0.026 amu
Energy released = 0.026 × 931 MeV
= 0.026 × 931 × 1.6 × 10-13 J
= 3.87 × 10-12 J
This is the energy produced by the consumption of three deutron atoms.
∴ Total energy released by 1040 deutrons
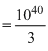 × 3.87 × 1012 J = 1.29 × 1028 J
× 3.87 × 1012 J = 1.29 × 1028 J
The average power radiated is P = 1016 W or 1016 J/s.
Therefore, total time to exhaust all deutrons of the star will be
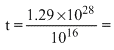 1.29 × 1012s
1.29 × 1012s  1012s
1012s
1H2 + 1H2 → 1H3 + p
1H2 + 1H3 → 2He4 + n
3 1H2 → 2He4 + n + p
Mass defect
ᐃm = (3 × 2.014 - 4.001 - 1.007 - 1.008) amu
= 0.026 amu
Energy released = 0.026 × 931 MeV
= 0.026 × 931 × 1.6 × 10-13 J
= 3.87 × 10-12 J
This is the energy produced by the consumption of three deutron atoms.
∴ Total energy released by 1040 deutrons
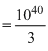 × 3.87 × 1012 J = 1.29 × 1028 J
× 3.87 × 1012 J = 1.29 × 1028 J The average power radiated is P = 1016 W or 1016 J/s.
Therefore, total time to exhaust all deutrons of the star will be
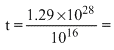 1.29 × 1012s
1.29 × 1012s  1012s
1012s Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The half life of a radioactive substance against α - decay is 1.2 × 107 s. What is the decay rate for 4.0 ...The wavelengths involved in the spectrum of deuterium (12D) are slightly different from that of hydrogen spectrum, becau...An electron from various excited states of hydrogen atom emit radiation to come to the ground state. Let λn, &...Read following statements carefully :-(a) The nucleus doesn′t have electron (b) The nucleus doesn′t emit an ...When an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the ...