ThermodynamicsHard
Question
1 mol of NH3 gas at 27oC is expanded under adiabatic condition to make volume 8 times (g = 1.33). Final temperature and work done, respectively, are
Options
A.150 K, 900 cal
B.150 K, 400 cal
C.250 K, 1000 cal
D.200 K, 800 cal
Solution
T2 = T1 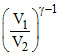 = 150 K
= 150 K
w = - CVDT = - CV(T2 - T1)
= -3 × 2 × (150 - 300)
= 900 cal
w = - CVDT = - CV(T2 - T1)
= -3 × 2 × (150 - 300)
= 900 cal
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
In which of the following reactions, standard reaction entropy change (ᐃSo) is positive and standardGibb′s e...If all degree of freedom of a three- dimensional N-atomic gaseous molecules is excited, then CP/CV ratio of gas should b...A reversible heat engine absorbs 40 kJ of heat at 500 K and performs 10 kJ of work rejecting the remaining amount to the...If the enthalpy change for the transition of liquid water to steam is 30 kJ mol-1 at 27oC, the entropy change for the pr...In a certain polytropic process, the volume of argon was increased 4 times. Simultaneously, the pressure decreased 8 tim...