ThermodynamicsHard
Question
In which of the following reactions, standard reaction entropy change (ᐃSo) is positive and standardGibb′s energy change (ᐃGo) decreases sharply with increasing temperature?
Options
A.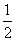 C graphite +
C graphite + 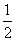 O2(g) →
O2(g) → 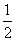 CO2(g)
CO2(g)
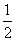 C graphite +
C graphite + 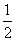 O2(g) →
O2(g) → 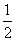 CO2(g)
CO2(g)B.C graphite + 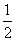 O2(g) → CO(g)
O2(g) → CO(g)
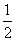 O2(g) → CO(g)
O2(g) → CO(g)C.CO(g) + 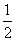 O2(g) → CO2(g)
O2(g) → CO2(g)
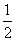 O2(g) → CO2(g)
O2(g) → CO2(g)D.Mg(s) + 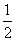 O2(g) → MgO(s)
O2(g) → MgO(s)
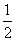 O2(g) → MgO(s)
O2(g) → MgO(s)More Thermodynamics Questions
A thermodynamic process is shown in the following figure. The pressure and volumes corresponding to some points in the f...Standard enthalpy of vaporisation ᐃvapHo for water at 100oC is 40.66 kJ mol-1. The internal energy of vaporisation...Which of the following pair does show the extensive properties?...In the reversible adiabatic expansion of an ideal monoatomic gas, the final volume is 8 times the initial volume. The ra...A sample of perfect gas that initially occupies 15.0 L at 300 K and 1.0 bar is compressed isothermally. To what volume m...