ThermodynamicsHard
Question
If the enthalpy change for the transition of liquid water to steam is 30 kJ mol-1 at 27oC, the entropy change for the process would be
Options
A.1.0 J mol-1 K-1
B.1.0 J mol-1 K-1
C.100 J mol-1 K-1
D.10 J mol-1 K-1
Solution
ᐃGo = ᐃHo - TᐃSo
Given, ᐃHvap = 30 kJ mol-1
ᐃGo = 0 at equilibrium,
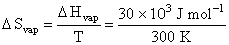
= 100 J mol-1 K-1
Given, ᐃHvap = 30 kJ mol-1
ᐃGo = 0 at equilibrium,
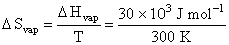
= 100 J mol-1 K-1
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Two moles of an ideal monoatomic gas undergoes a cyclic process ABCA as shown in the figure. What is the ratio of temper...If the ratio Cp/Cv = $\gamma$, the change in internal energy of the mass of a gas, when volume changes from V to 2V at c...An ideal gas expands in volume from 1 × 10-3 m3 to 1 × 10-2 m3 at 300 K against a constant pressure of 1 ×...The solubility of NaCl(s) in water at 298 K is about 6 moles per litre. Suppose you add 1 mole of NaCl(s) to a litre of ...Five moles of an ideal gas expand isothermally and reversibly from an initial pressure of 100 atm to a final pressure of...