Mole ConceptHard
Question
10 mL of a gaseous hydrocarbon is exploded with 100 mL of oxygen. The residual gas on cooling is found tp measure 95 mL of which 20 mL is absorbed by caustic soda and the remainder by alkaline pyrogallol. The formula of the hydrocarbon is
Options
A.CH4
B.C2H6
C.C2H4
D.C2H2
Solution
CxHy + 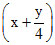 O2 → xCO2 +
O2 → xCO2 + 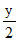 H2O
H2O
1 mL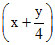 mL x mL
mL x mL 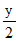 mL
mL
10 mL 10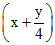 mL 10 x mL
mL 10 x mL
Volume absorbed by KOH = Volume of CO2
Volume of CO2 = 20 mL
∴ 10x = 20
x = 2 mL = 2 mol
volume obsorbed by alkaline pyrogallol = volume of O2 = 95 - 20 = 75 mL
Excess of O2 = 75 mL
Total O2 = 100 mL
Volume of O2 reacted = 100 - 75 = 25 mL
∴ 10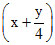 = 25 mL
= 25 mL
∴ y = 2
Formula of hydrocarbon = C2H2
1 mL
10 mL 10
Volume absorbed by KOH = Volume of CO2
Volume of CO2 = 20 mL
∴ 10x = 20
x = 2 mL = 2 mol
volume obsorbed by alkaline pyrogallol = volume of O2 = 95 - 20 = 75 mL
Excess of O2 = 75 mL
Total O2 = 100 mL
Volume of O2 reacted = 100 - 75 = 25 mL
∴ 10
∴ y = 2
Formula of hydrocarbon = C2H2
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
If 240 g of carbon is taken in a container to convert it completely to CO2 but in industry it has been found that 280 g ...The boiling point of 0.2 mol kg-1 solution of X in water is greater than equimolal solution of Y in water. Which one of ...Vapour pressure of chloroform (CHCl3) and dichloromethane (CH2Cl2) at 25oC are 200 mm Hg and 41.5 mm Hg respectively. Va...Two bulbs A and B contains 16 g O2 and 16g O3, respectively. Which of the statements are true ?...Calculate the milimoles of SeO32- in solution on the basis of following data : 70ml of solution of KBrO3 was back titate...