Mole ConceptHard
Question
Two bulbs A and B contains 16 g O2 and 16g O3, respectively. Which of the statements are true ?
Options
A.Both bulbs contain same number of atoms.
B.Both bulbs contain different number of atoms.
C.Both bulbs contain same number of molecules.
D.Bulb A contains NA/2 molecules while bulb B contains NA/3 molecules. (NA = Avogadro′s number).
Solution
Number of O2 atoms = 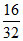 × 2 × NA = 1 × NAa. Number of O2 atoms =
× 2 × NA = 1 × NAa. Number of O2 atoms = 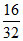 × 2 × NA = 1 × NA
× 2 × NA = 1 × NA
b. Number of O3 atoms =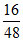 × 3 × NA = 1 × NA
× 3 × NA = 1 × NA
c. i. Number of O2 molecules =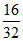 × NA =
× NA = 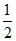 NA
NA
ii. Number of O2 molecules =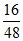 × NA =
× NA = 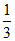 NA
NA
d. is correct
b. Number of O3 atoms =
c. i. Number of O2 molecules =
ii. Number of O2 molecules =
d. is correct
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
A quantity of 2.0 g of a triatomic gaseous element was found to occupy a volume of 448 ml at 76 cm of Hg and 273 K. The ...‘n’ mol of N2 and 0.05 mol of Ar is enclosed in a vessel of capacity 6 L at 1 atm and 27°C. The value of ‘n’ is(R = 0.08...A quantity of 720 g water is added in 230 g ethanol at a certain temperature to get 1 L of solution. Which of the follow...One molecule of haemoglobin will combine with four molecules of oxygen. If 1.0 g of haemoglobin combines with 1.642 ml o...While resting, an average 70 kg human male consumes 16.628 L of oxygen per hour at 270C and 100 kPa. How many moles of o...