Mole ConceptHard
Question
Calculate the milimoles of SeO32- in solution on the basis of following data :
70ml of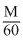 solution of KBrO3 was back titated with 12.5 mL of
solution of KBrO3 was back titated with 12.5 mL of 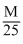 solution of 2 NaAsO2
solution of 2 NaAsO2
The reaction are given below.
I. SeO32- + BrO3- + H+ → SeO42-Br2 + H2O
II. BrO3- + AsO2- + H2O → Br- + AsO43- + H+
70ml of
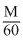 solution of KBrO3 was back titated with 12.5 mL of
solution of KBrO3 was back titated with 12.5 mL of 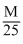 solution of 2 NaAsO2
solution of 2 NaAsO2The reaction are given below.
I. SeO32- + BrO3- + H+ → SeO42-Br2 + H2O
II. BrO3- + AsO2- + H2O → Br- + AsO43- + H+
Options
A.1.6 × 10-3
B.1.25
C.2.5 × 10-3
D.None of these
Solution
(I) 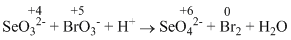
(II)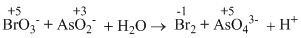
In reaction (II)
gm.eq. of BrO3- = gm.eq.of AsO2-
nBrO3- × 6 = nAsO2- × 2
=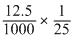 × 2 = 10-3
× 2 = 10-3
nBrO3- =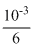
In reaction (I)
moles of BrO3- consumed
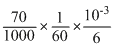 = 10-3
= 10-3
gm eq. of SeO32- = gm. eq. of BrO3-
nSeO32- × 2 = 10-3 × 5;
nSeO32- = 2.5 × 10-3
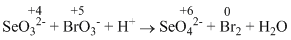
(II)
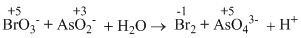
In reaction (II)
gm.eq. of BrO3- = gm.eq.of AsO2-
nBrO3- × 6 = nAsO2- × 2
=
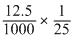 × 2 = 10-3
× 2 = 10-3nBrO3- =
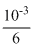
In reaction (I)
moles of BrO3- consumed
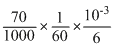 = 10-3
= 10-3gm eq. of SeO32- = gm. eq. of BrO3-
nSeO32- × 2 = 10-3 × 5;
nSeO32- = 2.5 × 10-3
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Which of the following is the incorrect conclusion regarding the following reaction?2 H2(g) + O2(g) → 2 H2O(l)...When a mixture consisting of 10 moles of SO2 and 16 moles of O2 were passed over a catalyst, 8 mole of SO3 were fomed at...At 373 K and 1 atm, if the density of liquid water is 1.0 g/ml and that of water vapour is 0.0006 g/ml, then the volume ...If 0.50 mole of BaCl2 is mixed with 0.20 mole of Na3 PO4, the maximum number of moles of Ba3(PO4)2 that can be formed is...The molecular formula of a compound is X4O9. If the compound contains 40% X by mass, then what is the atomic mass of X?...