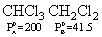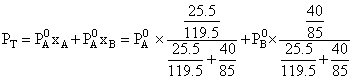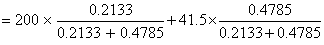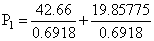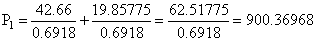Mole ConceptHard
Question
Vapour pressure of chloroform (CHCl3) and dichloromethane (CH2Cl2) at 25oC are 200 mm Hg and 41.5 mm Hg respectively. Vapour pressure of the solution obtained by mixing 25.5 g of CHCl3 and 40 g of CH2Cl2 at the same temperature will be:
(Molecular mass of CHCl3 = 119.5 u and molecular mass of CH2Cl2 = 85 u)
(Molecular mass of CHCl3 = 119.5 u and molecular mass of CH2Cl2 = 85 u)
Options
A.285.5 mm Hg
B.173.9 mm Hg
C.615.0 mm Hg
D.347.9 mm Hg
More Mole Concept Questions
In a hydrocarbon, mass ratio of hydrogen and carbon is 1 : 3, The empirical formula of hydrocarbon is...In the atomic weight determination, Dalton suggested the formula of water as HO and the composition of water as hydrogen...Consider the following statements1. If all the reactants are not taken in their stoichiometric ratio, then at least one ...The percentage of Fe(III) present in iron ore Fe0.93 O1.00 is (Fe = 56)...The mercury content of a stream was believed to be above the minimum considered safe limit (1 part per billion, by mass)...