Mole ConceptHard
Question
If 240 g of carbon is taken in a container to convert it completely to CO2 but in industry it has been found that 280 g of CO was also formed along with CO2. Find the mole percentage yield of CO2. The reactions occurring are C + O2 → CO2 ; C + 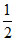 O2 → CO
O2 → CO
Options
A.25 %
B.50 %
C.75 %
D.100%
Solution
C + O2 → CO2 + CO
POAC on ′C′ atom
1 (mole of C) = 1 (mole of CO2) + 1 (mole of CO)
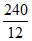 = mole of CO2 +
= mole of CO2 + 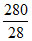
Mole of CO2 = 20 - 10 = 10
Mole % of CO2 =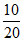 ×100 = 50%.
×100 = 50%.
POAC on ′C′ atom
1 (mole of C) = 1 (mole of CO2) + 1 (mole of CO)
Mole of CO2 = 20 - 10 = 10
Mole % of CO2 =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
At $T(K),100\text{ }g$ of $98\% H_{2}{SO}_{4}(w/w)$ aqueous solution is mixed with 100 g of $49\% H_{2}{SO}_{4}(w/w)$ aq...If 0.50 mole of BaCl2 is mixed with 0.20 mole of Na3 PO4, the maximum number of moles of Ba3(PO4)2 that can be formed is...Law of multiple proportions is not applicable for the oxide(s) of...When a sample of hydrogen fluoride is cooled to 303 K, most of the molecules undergo dimerization. If the vapour density...When burnt in air, a 12.0 g mixture of carbon and sulphur yields a mixture of CO2 and SO2, in which the number of moles ...